Plasma Systems | Cleaning & Surface Treatment | AST Products
Plasma Applications
AST Products, Inc. is experienced in system design and process development for special plasma surface treatment applications. Plasma processes have been developed to attain a variety of specific surface properties. Below are some examples of common plasma applications.
Plasma Process Examples:
Surface Cleaning
Oxygen plasma treatment is a safe and environmentally friendly alternative to traditional cleaning methods. A wide variety of industries utilize gas plasma treatment to remove organic surface contamination from materials that require critical cleaning. The active species in the oxygen combined with VUV energy creates a chemical reaction with the surface contaminants, resulting in their volatilization and removal from the reaction chamber. Applications include cleaning of circuit boards for improved wire bonding, removal of organic contamination from glass slides and flat panels, cleaning of medical devices and other materials such as various metals and ceramics.
Adhesion Promotion
Improving adhesion between two surfaces is a common application. Good adhesion requires strong interfacial forces via chemical compatibility and/ or chemical bonding. Plasma surface treatment can also assist in creating chemically active functional groups, such as amine, carbonyl, hydroxyl and carboxyl groups, to improve interfacial adhesion. Common applications include pretreatment for medical catheters, cardiovascular stents, syringe components, dialysis pump parts and plastic films for drug bags. Plasma is also used to improve bondability on substrates, such as glass, polymers, ceramics and various metals.
Controlling Surface Energies
Plasma processing can also be used to tailor surface energies. Hydrophilic and hydrophobic surfaces can be created on polymers through interaction with a gas plasma. Using oxygen to create hydroxil functionality increases the wettability of a surface. This process has been used to enhance the performance of a catheter by creating a wettable surface on the polymer tubing. Similarly, surfaces can be specifically engineered to modify protein binding and improve blood compatibility. Common applications include tissue culture wares, filtration or separation media and contact lenses.
Improving Biocompatibility
Biomaterials that come in contact with blood or protein require special surface treatments to enhance biocompatibility. Amine functional groups, which are attached by ammonia plasma treatment, act as hooks for anticoagulants, such as heparin, and thereby decrease thrombogenicity. Synthetic polymer implant materials can be surface activated using radio frequency plasma techniques to enable covalent immobilization of cell binding peptides derived from the extra-cellular matrix proteins: fibronectin and laminin. The resulting grafted peptides can promote complete coverage of a surface with a monolayer of intact, healthy endothelial cells to form a natural, blood compatible surface. The endothelialized bioimplants will have improved biocompatibility and reduced antigenicity and thrombogenicity. Tetraflouroethylene plasma coatings on small-diameter Dacron vascular grafts have demonstrated dramatic improvements in patency and decreased embolization.
Enhancing Performance
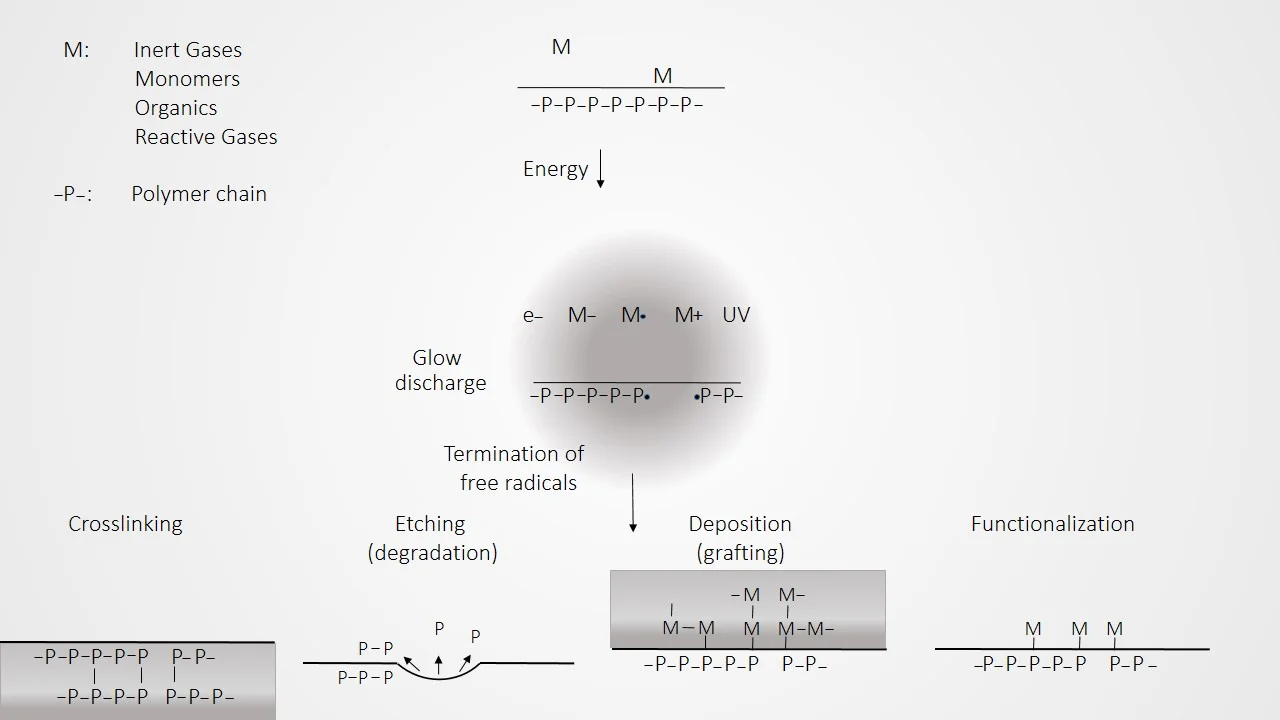
Surface crosslinking is often used to enhance the performance of polymers. The activity of the plasma creates a higher crosslinking density within the material to depths of a few thousand angstroms. The resulting increase in hardness and chemical resistance can enhance performance in many applications. Here are some examples:
Silicone rubber components treated in inert gas plasma can be modified to form a hard “skin” on the surface. The result is a substantial decrease in surface tack and coefficient of friction.
By using a plasma immobilization process precoated molecules can be directly crosslinked onto polymer surfaces. The molecules immobilized by this process can be organic compounds, surfactants, polymers or proteins, and do not require unsaturated double bonds in the molecules.
Plasma polymers have also been used in conjunction with parylene coating to provide tough barriers that strongly adhere to the substrate. There are many other types of coatings that can be similarly enhanced.
What is Plasma?
Plasma can be defined as a partially or wholly ionized gas with a roughly equal number of positively and negatively charged particles. Some scientists have dubbed plasma the “fourth state of matter” because while plasma is neither gas nor liquid, its properties are similar to those of both gases and liquids.
There are two types of plasma – high temperature and low temperature. A good example of naturally occurring high temperature plasma is lightning. This type of plasma can be artificially generated using a high voltage, high temperature arc, which is the basis for the corona discharge process and for the plasma torch used to vaporize and redeposit metals.
Low temperature plasmas, used in surface modification and organic cleaning, are ionized gases generated at pressures between 0.1 and 2 torr. These types of plasmas work within a vacuum chamber where atmospheric gases have been evacuated typically below 0.1 torr. Low pressure allows for a relatively long free path of accelerated electrons and ions. Since the ions and neutral particles are at or near ambient temperatures and the long free path of electrons, which are at high temperature or electron volt levels, have relatively few collisions with molecules at this pressure the reaction remains at low temperature.
How Does Plasma Surface Modification Work?
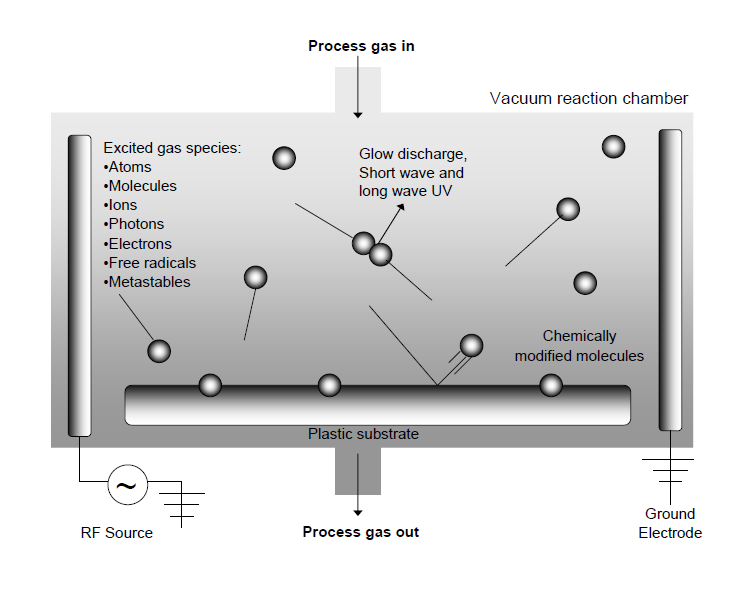
In the plasma surface modification process, a glow discharge plasma is created by evacuating a reaction chamber and then refilling it with a low-pressure gas. The gas is then energized by one of the following types of energy: radio frequency, microwaves, and alternating or direct current. The energetic species in gas plasma include ions, electrons, radicals, metastables, and photons in the short-wave ultraviolet (UV) range. Surfaces in contact with the gas plasma are bombarded by these energetic species and their energy is transferred from the plasma to the solid. These energy transfers are dissipated within the solid by a variety of chemical and physical processes (as schematically illustrated in figure 3) to result in a unique type of surface modification that reacts with surfaces in depths from several hundred angstroms to 10µm without changing the bulk properties of the material.
A wide variety of parameters can greatly affect the physical characteristics of a plasma and subsequently affect the surface chemistry obtained by plasma modification. Processing parameters, such as gas types, treatment power, treatment time and operating pressure, can be varied by the user; however system parameters, such as electrode location, reactor design, gas inlets and vacuum are set by the design of the plasma equipment. This broad range of parameters offers greater control over the plasma process than that offered by most high-energy radiation processes.
Other advantages of plasma treatment include low environmental impact, no line-of-sight problem as compared to laser and UV radiation, and a pinhole-free coating. In addition, although gas plasma treatments are typically carried out in batch processes, continuous on-line treatment of fibers, tubing, membranes, fabrics and films is also possible.
Biomedical Applications of Plasma Surface Modification
Although many synthetic biomaterials have physical properties that meet and even exceed those of natural body tissue, they can often cause adverse physiological reactions such as infection, inflammation and thrombosis formation. Through surface modification, biocompatibility as well as biofunctionality can be achieved without changing the bulk properties of the material. There are many ways by which to alter the interaction of biomaterials with their physiological environments, of these, plasma surface modification provides device manufacturers with a flexible, safe and environmentally friendly process that is extremely effective.
Biomedical Application Examples
Bioseparation
Polymer membranes with selective permeation are widely used for applications such as, haemodialysis, protein purification, artificial organs and biosensors. Membrane materials must exhibit high selectivity of permeation to achieve high product purity while retaining high permeation rates to maximize productivity. High selectivity can be enhanced by incorporating chemical functionality (grafting) or physical restriction (controlled pore size) into the membranes.
Sterilization
Plasma technology has also been considered for disinfection and sterilization of medical devices. The most convenient aspect of plasma technology is the potential for simultaneous surface modification and sterilization in biomedical device fabrication. Plasma sterilization may be suitable for medical implants and devices that are sensitive to temperature, radiation and chemicals.
Ocular Prostheses
Products such as contact lenses and intraocular lenses have been successfully modified by plasma treatment to impart protein- and cell-repelling characteristics, decrease bacterial adhesion, improve wettability and enhance patient comfort. Also, an ammonia plasma treatment has been applied to artificial corneas to enhance corneal epithelial cell attachment and growth.
Orthopaedic Applications
Plasma has also been employed to modify the surface of metal implants for adhesion promotion to bone cements or to enhance cell attachment and growth. Plasma crosslinking of a bioabsorbable polymer surface has been shown to regulate the material’s degradation rate. Plasma technology can also improve the mechanical properties of totally absorbable composite bone plates strengthening the interfacial bonding between reinforced absorbable fibers and the absorbable matrix, and by retarding water penetration into the reinforced fibers and matrix to slow the rate of degradation.
Tissue Culturing
The surfaces of culture substrates, such as petri dishes, roller bottles, microcarriers and membranes can be modified by plasma treatment to improve cell growth, protein binding or non-binding and cell-specific attachment by controlling surface chemical structures, surface energies and/or surface charge states.